Unit Questions: (what you should be able to answer by the end of the unit)
- How do temperature, pressure, and volume affect gases in a closed system?
- What are the gas laws and how do they explain the effects of temperature, pressure, and volume on gases?
- How does the kinetic molecular theory explain gas behavior?
- Why some gases are not considered ideal gases?
- How are gases involved in a chemical reaction quantified?
- How are various forms of energy categorized?
- How are energy changes in a chemical reaction and matter quantified?
Graphics: |
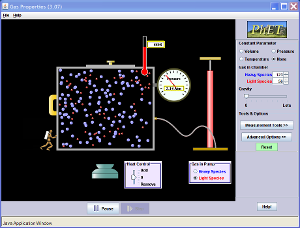
Simulations:Click on the button below for a cool simulation/calculation:
|
If I were studying for the gas law test, I would check out all the extra worksheets/notes/videos below. I would also make sure I could handle these type of questions:
1. A gas is at 1.33 atm of pressure and a volume of 66L. What will the pressure be if the volume is reduced to 0.419 L?
____________
2. Which of these changes would NOT cause an increase in the pressure of a gaseous system?
A) Additional amounts of the same gas are added to the container.
B) Another gas is added to the container.
C) The temperature is increased.
D) The container is made larger.
3. A gas storage tank is a 172kPa and 35oC. What temperature is the gas at if the pressure increases to 2.00 atm?
as well as these type:
A breathing mixture used by deep-sea divers contains helium, oxygen, and carbon dioxide. What is the partial pressure of oxygen at 101 kPa if PHe = 96.1 kPa and PCO2= 0.6 kPa?
A sample of gas occupies 40.0L at -123°C. What volume does the sample occupy at 27°C?
The tendency of molecules to move toward areas of lower concentration is called _____.
The volume of a gas is reduced from 4 L to 0.5 L while the temperature is held constant. How does the gas pressure change?
A) increases by a factor of four
B) increases by a factor of eight
C) decreases by a factor of eight
D) increases by a factor of two
1. A gas is at 1.33 atm of pressure and a volume of 66L. What will the pressure be if the volume is reduced to 0.419 L?
____________
2. Which of these changes would NOT cause an increase in the pressure of a gaseous system?
A) Additional amounts of the same gas are added to the container.
B) Another gas is added to the container.
C) The temperature is increased.
D) The container is made larger.
3. A gas storage tank is a 172kPa and 35oC. What temperature is the gas at if the pressure increases to 2.00 atm?
as well as these type:
A breathing mixture used by deep-sea divers contains helium, oxygen, and carbon dioxide. What is the partial pressure of oxygen at 101 kPa if PHe = 96.1 kPa and PCO2= 0.6 kPa?
A sample of gas occupies 40.0L at -123°C. What volume does the sample occupy at 27°C?
The tendency of molecules to move toward areas of lower concentration is called _____.
The volume of a gas is reduced from 4 L to 0.5 L while the temperature is held constant. How does the gas pressure change?
A) increases by a factor of four
B) increases by a factor of eight
C) decreases by a factor of eight
D) increases by a factor of two
Notes:This lady does GREAT videos:
Purdue University: http://chemed.chem.purdue.edu/genchem/topicreview/bp/ch4/gaslaws3.html
Articles:
|
POGIL:
Worksheets:
Animations: |
Labs:mini/demos:
Full:
Collapse the Can :
http://www.nsta.org/publications/news/story.aspx?id=51859#Anchor-49575 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Video Notes:
Mr. Guthrie: